Children’s National Hospital joins the Mendelian Genomics Research Consortium, receiving $12.8 million

Dr. Eric Vilain accompanied by a fellow researcher at the new Research & Innovation Campus.
Children’s National Hospital announces a $12.8 million award from the National Institutes of Health’s National Human Genome Research Institute (NHGRI) to establish the only Pediatric Mendelian Genomics Research Center (PMGRC) as part of a new Mendelian Genomics Research Consortium. Researchers at Children’s National and Invitae — a leading medical genetics company — will identify novel causes of rare inherited diseases, investigate the mechanisms of undiagnosed conditions, enhance data sharing, and generally interrogate Mendelian phenotypes, which are conditions that run in families.
“Our overall approach provides an efficient and direct path for pediatric patients affected with undiagnosed inherited conditions through a combination of innovative approaches, allowing individuals, families and health care providers to improve the management of the disease,” says Eric Vilain, M.D., Ph.D., director of the Center for Genetic Medicine Research at Children’s National.
To accelerate gene discovery for Mendelian phenotypes and the clinical implementation of diagnosis, the consortium will leverage the broad pediatric clinical and research expertise of the Children’s National Research Institute and laboratories in partnership with Invitae. The Molecular Diagnostics Laboratory at Children’s National will provide genetic testing for patients in the Washington, D.C., metropolitan area. Invitae will provide genetic testing for patients from elsewhere in the U.S., giving the project a national reach and allowing researchers to leverage more robust data. Integrative analyses will be performed jointly with scientists at Children’s National and Invitae.
“Some patients have genetic test results that are ‘negative,’ meaning the results do not explain their condition. When a patient receives a negative result, it is challenging for parents and doctors to know what to do next,” says Meghan Delaney, D.O., M.P.H., chief of the Division of Pathology and Laboratory Medicine and Molecular Diagnostics Laboratory at Children’s National. “The project will provide an avenue to possibly find an explanation of their child’s condition. Besides filling an important clinical gap, the results will add new knowledge for future patients and the scientific community.”
“Too often parents of children suffering from a rare condition find themselves in a protracted diagnostic odyssey when early intervention could mean better overall outcomes,” says Robert Nussbaum, M.D., chief medical officer of Invitae. “We are proud to partner with Children’s National Research Institute on this important effort to identify the genetic cause of these rare conditions earlier and improve the chances that children with such conditions can receive the appropriate treatments and live healthier lives.”
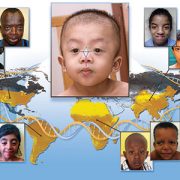
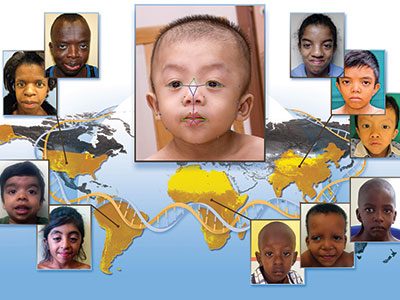
Deciphering Mendelian conditions will help diagnose more of the estimated 7,000 rare inherited diseases and predict the tremendous variability of clinical presentations in both rare and common conditions caused by the same gene.
There is also a need to establish a new standard of care to bridge the gap in the use of genomic information from diagnosis to improved outcomes. The consortium will establish best practices for obtaining a genetic diagnosis, offering an explanation for the condition to affected patients, and is likely to provide additional explanations for basic biological mechanisms, increasing the knowledge of physiopathology and possibly leading to better condition management.
The PMGRC will enroll an average of 2,600 participants per year with suspected Mendelian phenotypes and previously non-diagnostic tests and their family members. The integration of multiple genomic technologies, including short and long read genome sequencing, optical genome mapping and RNA-sequencing, will enable these discoveries. To disambiguate uncertain variants and candidate genes, the PMGRC will use whole transcriptome analysis, RNA-sequencing, CRE-sequencing and functional modeling.
Since many Mendelian conditions first appear prenatally or during infancy, Children’s National will have a unique bed-to-bench-to-bed symbiosis. Patients eligible for the study will come from across the multiple specialty divisions of Children’s National, including the Children’s National Rare Disease Institute, and nationally through the partnership with Invitae. From there, experts from the Children’s National Center for Genetic Medicine Research will enroll patients and integrate the initial clinical test results with broad-based genomic interrogation, leading to new diagnoses and novel discoveries. Finally, the results will be verified and returned to clinicians, which will help inform targeted therapies.
Typically, the patients eligible for this study jump from specialist to specialist without an answer, have a condition that appears in other family members or they have symptoms involving more than one affected organ, which suggests a complex developmental condition. The PMGRC at Children’s National will help find answers to the causes of many puzzling pediatric conditions, providing faster clinical diagnoses and opening up pathways to potentially better treatments.
Dr. Vilain’s work will be based at the Children’s National Research & Innovation Campus on the grounds of the former Walter Reed Army Medical Center in Washington, D.C. The campus is also home to the Children’s National Rare Disease institute — one of the largest clinical genetics program in the United State that provides care to more than 8,500 rare disease patients.