New study finds prenatal opioid exposure linked to smaller newborn brain volumes
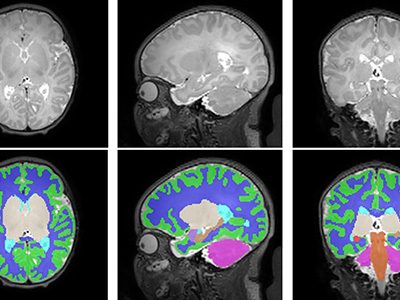 A new study out of the Center for Prenatal, Neonatal & Maternal Health Research, directed by Catherine Limperopoulos, PhD, led by Yao Wu, PhD and Stephanie Merhar, MD, MS, out of Cincinnati Children’s Hospital Medical Center, sought answers to the question: “Do brain volumes differ in opioid-exposed vs. unexposed newborns?” In one of the largest studies of this kind, researchers found that prenatal exposure to opioids is associated with smaller brain volumes in newborns. These findings from the landmark Outcomes of Babies with Opioid Exposure (OBOE) study build on, reinforce smaller studies, and achieve a better understanding of the impacts of prenatal opioid exposure (POE).
A new study out of the Center for Prenatal, Neonatal & Maternal Health Research, directed by Catherine Limperopoulos, PhD, led by Yao Wu, PhD and Stephanie Merhar, MD, MS, out of Cincinnati Children’s Hospital Medical Center, sought answers to the question: “Do brain volumes differ in opioid-exposed vs. unexposed newborns?” In one of the largest studies of this kind, researchers found that prenatal exposure to opioids is associated with smaller brain volumes in newborns. These findings from the landmark Outcomes of Babies with Opioid Exposure (OBOE) study build on, reinforce smaller studies, and achieve a better understanding of the impacts of prenatal opioid exposure (POE).
Dive Deeper
The Advancing Clinical Trials in Neonatal Opioid Withdrawal (ACT-NOW)’s OBOE study is a multi-site observational study of newborns with prenatal opioid exposure and a control group of unexposed newborns from four different sites in the United States – Case Western Reserve University, Cincinnati Children’s Hospital Medical Center, University of Alabama at Birmingham, and Children’s Hospital of Philadelphia.
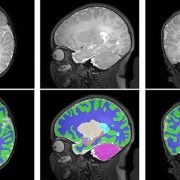
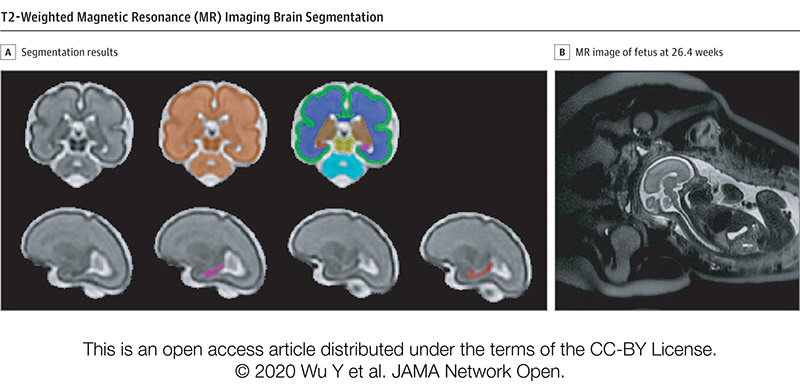
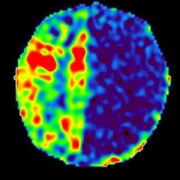
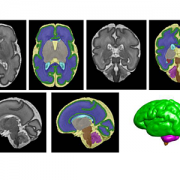
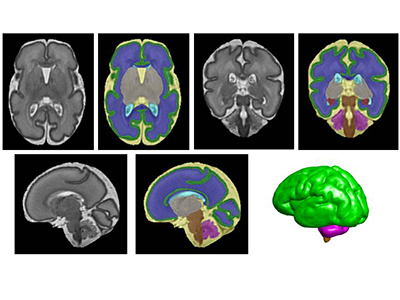
In a study involving 173 newborns who were exposed to opioids during pregnancy and 96 newborns not exposed to opioids prenatally showed smaller brain sizes in several key areas. Specifically, these exposed babies had smaller total brain volumes, as shown through MRI, as well as reduced volumes in important parts of the brain, including the cortex (outer layer of the brain), deep gray matter (areas that control movement and emotions), white matter (which helps transmit signals in the brain), cerebellum (responsible for coordination and movement), brainstem (controls basic functions like breathing), and the amygdala (involved in emotions and memory).
Further details showed that newborns exposed to medication for opioid use disorder (MOUD) during pregnancy with methadone, had smaller white matter volumes, while those exposed to MOUD with buprenorphine had smaller volumes specifically in the right amygdala. Additionally, newborns who were exposed to opioids plus additional substances such as THC and gabapentin had smaller volumes in even more brain areas compared to those who were only exposed to opioids.
What’s Next
The OBOE study sets the groundwork for further research into the long-term impact of opioid exposure during pregnancy. Additional work is necessary to expand on these findings and how they relate to functions in childhood – including exploring the way these reduced brain volumes may impact cognitive, behavioral, and motor impairments. The study raises important questions about how current guidelines for MOUD during pregnancy – specifically with methadone and buprenorphine – might evolve considering these findings. This study highlights the need for further research to assess the long-term effects of MOUD regimens on both maternal and infant outcomes.
This significant study underscores the importance of multi-disciplinary collaboration in opioid exposure research, effective regulation, and policy interventions – involving healthcare providers, researchers, policymakers, and affected families – to best mitigate the consequences and improve the health outcomes of children affected by prenatal opioid exposure.
You can read the full study, Antenatal Opioid Exposure and Global and Regional Brain Volumes in Newborns, and its companion editorial, Following the Developing Brain Affected by Opioid Exposure, in the Journal of the American Medical Association.
Additional authors from Children’s National include Kushal Kapse, BS, MS, and Josepheen De Asis-Cruz, MD, PhD. Other authors include Carla M. Bann, PhD, Jamie E. Newman, PhD4, Nicole Mack, MS, Sara B. De Mauro, MD, MSCE, Namasivayam Ambalavanan, MD, Jonathan M. Davis, MD, Scott A. Lorch, MD, MSCE5, Deanne Wilson-Costello, MD, Brenda B. Poindexter, MD and Myriam Peralta-Carcelen, MD.