Tracking preemies’ blood flow to monitor brain maturation
Blood is the conduit through which our cells receive much of what they need to grow and thrive. The nutrients and oxygen that cells require are transported by this liquid messenger. Getting adequate blood flow is especially important during the rapid growth of gestation and early childhood – particularly for the brain, the weight of which roughly triples during the last 13 weeks of a typical pregnancy. Any disruption to blood flow during this time could dramatically affect the development of this critical organ.
Now, a new study by Children’s National Health System researchers finds that blood flow to key regions of very premature infants’ brains is altered, providing an early warning sign of disturbed brain maturation well before such injury is visible on conventional imaging. The prospective, observational study was published online Dec. 4, 2017 by The Journal of Pediatrics.
“During the third trimester of pregnancy, the fetal brain undergoes an unprecedented growth spurt. To power that growth, cerebral blood flow increases and delivers the extra oxygen and nutrients needed to nurture normal brain development,” says Catherine Limperopoulos, Ph.D., director of the Developing Brain Research Laboratory at Children’s National and senior author of the study. “In full-term pregnancies, these critical brain structures mature inside the protective womb where the fetus can hear the mother and her heartbeat, which stimulates additional brain maturation. For infants born preterm, however, this essential maturation process happens in settings often stripped of such stimuli.”
The challenge: How to capture what goes right or wrong in the developing brains of these very fragile newborns? The researchers relied on arterial spin labeling (ASL) magnetic resonance (MR) imaging, a noninvasive technique that labels the water portion of blood to map how blood flows through infants’ brains in order to describe which regions do or do not receive adequate blood supply. The imaging work can be done without a contrast agent since water from arterial blood itself illuminates the path traveled by cerebral blood.
“In our study, very preterm infants had greater absolute cortical cerebral blood flow compared with full-term infants. Within regions, however, the insula (a region critical to experiencing emotion), anterior cingulate cortex (a region involved in cognitive processes) and auditory cortex (a region involved in processing sound) for preterm infants received a significantly decreased volume of blood, compared with full-term infants. For preterm infants, parenchymal brain injury and the need for cardiac vasopressor support both were correlated with decreased regional CBF,” Limperopoulos adds.
The team studied 98 preterm infants who were born June 2012 to December 2015, were younger than 32 gestational weeks at birth and who weighed less than 1,500 grams. They matched those preemies by gestational age with 104 infants who had been carried to term. The brain MRIs were performed as the infants slept.
Blood flows where it is needed most with areas of the brain that are used more heavily commandeering more oxygen and nutrients. Thus, during brain development, CBF is a good indicator of functional brain maturation since brain areas that are the most metabolically active need more blood.
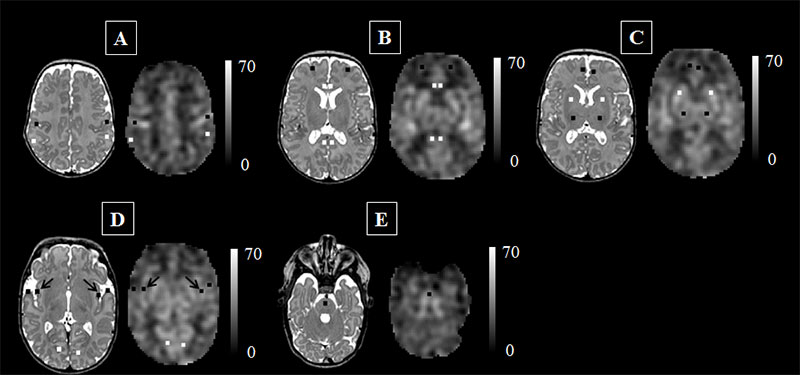
This figure represents the cerebral blood flow (CBF) maps, corresponding anatomical image aligned to the CBF map, and the regions of interest examined. The scale indicates the quantitative value of the CBF map and is expressed in mL/100g/min. The data are from a preterm infant scanned at term age without evidence of brain injury. The insula (see black arrows in panel ‘D’) may be particularly vulnerable to the added stresses of the preterm infant’s life outside the womb.
Credit: M. Bouyssi-Kobar, et al., The Journal of Pediatrics.
“The ongoing maturation of the newborn’s brain can be seen in the distribution pattern of cerebral blood flow, with the greatest volume of blood traveling to the brainstem and deep grey matter,” says Marine Bouyssi-Kobar, M.S., the study’s lead author. “Because of the sharp resolution provided by ASL-MR images, our study finds that in addition to the brainstem and deep grey matter, the insula and the areas of the brain responsible for sensory and motor functions are also among the most oxygenated regions. This underscores the critical importance of these brain regions in early brain development. In preterm infants, the insula may be particularly vulnerable to the added stresses of life outside the womb.”
Of note, compromised regional brain structures in adults are implicated in multiple neurodevelopmental disorders. “Altered development of the insula and anterior cingulate cortex in newborns may represent early warning signs of preterm infants at greater risk for long-term neurodevelopmental impairments,” Limperopoulos says.
Research reported in this post was supported by the Canadian Institutes of Health Research, MOP-81116; the SickKids Foundation, XG 06-069; and the National Institutes of Health under award number R01 HL116585-01.