Q&A with Daniel J. Licht, M.D.: The future of medicine is in light

A pediatric neurologist who specializes in children with congenital heart disease, Dr. Licht initially came to this area of research as he considered ways to ensure children’s brains have adequate oxygen delivery during heart care, preserving neurological health and improving long-term outcomes.
Daniel J. Licht, M.D., joins Children’s National Hospital with a vision: He believes non-invasive devices built using biomedical optics – or instruments using light – can give clinicians invaluable information about how the brain and other organs are functioning.
A pediatric neurologist who specializes in children with congenital heart disease, Dr. Licht initially came to this area of research as he considered ways to ensure children’s brains have adequate oxygen delivery during heart care, preserving neurological health and improving long-term outcomes. He sees countless applications for using the properties of light in pediatric medicine.
Dr. Licht, whose name coincidentally also means “light” in German, is planning to establish a program for biomedical optics at Children’s National, built on the pillars of education, innovation and commercialization. He wants to tap into the resources of the Sheik Zayed Institute of Pediatric Surgical Innovation and expertise across the hospital. He is launching this effort as part of the new Center for Prenatal, Neonatal & Maternal Health Research.
Q: How can light be used diagnostically?
A: I believe that light is truly the future of biomedical devices, especially in children. Light can penetrate human tissues deeply, whether it’s muscle, liver or kidney. For example, you can put a light at the end of an endoscope and someday do virtual biopsies. It’s all a matter of understanding the properties of light, and how to manipulate light to give you the answers that you need. The applications are truly infinite.
Q: What has your initial work in neurology shown?
A: One of the instruments that we have developed can measure cerebral blood flow and quantitatively show the oxygen use of the brain. That’s important because it’s easy to measure oxygen delivery, but it’s hard to balance supply-and-demand without knowing the patient’s unique demand. We now have preclinical data and information from about 500 patients.
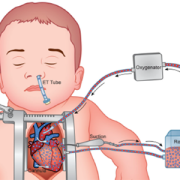
In terms of what’s ahead, many therapies today aren’t targeted to the individual, so Johnny’s brain-oxygen demand may not be the same as Sarah’s brain-oxygen demand, even if they both have congenital heart disease. As a patient waits for surgery, we also have found that the brain-oxygen demand increases, but if the demand is not met, this can lead to pre-operative brain injury. This technology could change the whole conversation about the timing of surgery. In addition, we can measure the brain-oxygen demand intraoperatively. We are finding that we can actually define the right perfusion strategy for each patient, rather than making uniform decisions for all patients with a shared diagnosis.
Lastly, beyond the operating room, we can use this technology for countless conditions. It would help with the treatment of almost any disease in the critical care unit, when we are using tools like ECMO (extracorporeal membrane oxygenation, a salvage technique), and we need to monitor a patient’s status. We can also use it to measure intracranial pressure. In very simple terms, if a child with a shunt comes into the emergency room with a headache, we can noninvasively measure the pressure and see how it’s changed without a head CT. We can decide who needs to go to the operating room – and who doesn’t – without radiation.
Q: How did your career bring you to this point?
A: My interest has always been in brain injury and kids with congenital heart disease. Years ago, I started out using MRI because it was the technology that was bright and shiny at the time. I was part of a team that developed an MRI sequence for measuring cerebral blood flow. We made some discoveries that indicated the culprit for brain injury was not the surgeries. Instead, there was something with the babies.
Unfortunately, with MRI, it’s a big, expensive instrument, and you have to take the baby to the machine for a single point-in-time measurement. So I started working with a physicist at the University of Pennsylvania to develop a way to measure the motion of particles, specifically red blood cells, to study cerebral blood flow. We found ways to use light, and this is what I hope to build and commercialize at Children’s National. By the end of my career, I hope to be able to say that we got this into clinical care.