Gene therapy’s slow rebirth

A speech by outgoing American Pediatric Society President Mark L. Batshaw, M.D., explored the impact of a single clinical trial on the entire field.
Gene therapy – delivering genetic material into patients’ cells as a way to treat or cure their diseases – has immense promise to alleviate or end many lifelong and deadly conditions. This treatment has so much potential that it was a heavy focus of research and research dollars around the world in the 1980s and 1990s.
However, many of these efforts came to a screeching halt in 1999 when a teenaged patient named Jesse Gelsinger died in a gene therapy trial aimed at curing a disease called ornithine transcarbamylase deficiency, a urea cycle disorder. Gelsinger’s death triggered a number of investigations, halted gene therapy trials in the United States, and severely restricted financial support from federal, foundation and industry funders.
The tragedy also spurred Mark L. Batshaw, M.D., one of the clinical trial investigators and newly named Chief Academic Officer at Children’s National Health System, to turn in his resignation. The chief executive at the time declined to accept it, instead naming an outside panel to investigate Dr. Batshaw’s role in a study marred by conflicts of interest, delays in updating patient consent forms, lack of adherence to the study protocol and ineffective team leadership.
As Dr. Batshaw passed the gavel to the next president of the American Pediatric Society during the Pediatric Academic Societies’ annual meeting this spring, he told attendees of his Presidential Address that “not a day goes by that I don’t think of Jesse Gelsinger and his family and hope that the work our team has continued will honor him by eventually achieving success with gene therapy.” In an act of altruism, 18-year-old Jesse Gelsinger had joined the trial with the aim of helping other kids suffering from metabolic disorder.
Dr. Batshaw recognizes that his is an unusual choice, speaking about his “greatest professional failure” when predecessors have used their addresses to speak exclusively about scientific accomplishments.
“Because I was a principal, I think telling this story first of all says, hey look, this guy who is president of this organization, who has had a significant career, is willing to talk openly about a failure and how he dealt with it and how the field dealt with that failure,” Dr. Batshaw says. “Secondly, the field of gene therapy right now is starting to explode. It’s telling two different stories in an integrated way: One is of a great personal failure – and failure of an entire field. And the recovery from that, and what the future will be for a technology that holds great promise.”
More than 1,000 gene therapy trials are currently underway, 23 of them at Phase III, the pivotal stage that makes or breaks approval by the Food and Drug Administration (FDA). Dr. Batshaw estimates about a dozen of those are likely to demonstrate robust enough results to progress to a formal application for FDA approval. “After a period of virtually no growth in gene therapy trials from 2000 to 2013, there has been a marked upswing in the past two to three years,” he says.
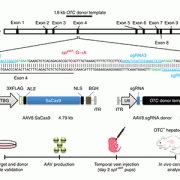
Children’s National is a study site for one of those clinical trials, a Phase I/II adenoassociated virus (AAV)-mediated gene therapy for late-onset ornithine transcarbamylase (OTC) deficiency. Children with urea cycle disorders have enzyme deficiencies that leave them unable to adequately dispose of waste nitrogen. Often as newborns, they develop severely elevated ammonia in their brains leading to encephalopathy, an often fatal condition. The Phase I work will test escalating doses in three patients for safety. The Phase II work will explore whether the gene therapy improves outcomes like lowering ammonia levels and improving patients’ ability to convert ammonia to urea. (A precursor study in an experimental model was among the most impactful research papers published by Children’s National authors in 2016.)
“So, for both our group’s program – and viral-delivered gene therapy in general – there has been a rebirth after the disastrous outcome of the initial adenovirus trial in OTC deficiency,” Dr. Batshaw said in his prepared remarks. “This resurgence has likely been fueled by improved viral vectors, especially AAVs, and an improved economy and industry investment. The future of gene therapy is likely to be enhanced by new genetic therapy platforms including RNA interference as a means of vertically transmitted gene regulation and the CRISPR gene-editing technology. It will also be impacted by the results of the trials that will be completed in the next few years, especially those using AAV vectors in hemophilia A and B, spinal muscular atrophy and leukemia.”
Looking forward 10 years, Dr. Batshaw is hopeful that gene therapy will become part of the therapeutic tools routinely used to help patients who suffer from rare disease and cancer. Making that next leap forward will be powered by innovative research, including work by colleagues at Children’s National. Among the presenters at PAS2017, the world’s largest pediatric research meeting, were more than 100 Children’s presenters, speakers and moderators.
“It makes me very proud that there are so many clinicians who are also scientists who are not satisfied with simply doing things the way they have always been doing it but constantly questioning how can we do things better for our children?” Dr. Batshaw says. “Our whole focus at Children’s National is caring for children, and that means caring for them the very best way possible and not being satisfied with current therapy if it’s not curative.”