DOD $1.3M award will launch clinical trial to treat sarcoma
MPNST is a type of cancer called a sarcoma.
The Department of Defense (DOD) awarded Children’s National Hospital $1.3M to launch a unique clinical trial. The trial will evaluate the safety of a novel drug for patients with a rare but aggressive cancer known as malignant peripheral nerve sheath tumors (MPNST).
MPNST is a type of cancer called a sarcoma. While rare in the general population, about half of all MPNST are diagnosed in people with Neurofibromatosis Type 1 (NF1), a condition characterized by changes in skin coloring.
“MPNST is a life-threatening cancer for which there are no adequate medical options,” said AeRang Kim, M.D., Ph.D., director of clinical research of the Division of Oncology at Children’s National. “With the support of this grant, we will conduct a clinical trial to identify effective agents that could be of great benefit to all patients with NF1 who are at risk for sarcoma.”
MPNST are aggressive and frequently metastasize. The tumors that are not able to be removed with surgery rapidly progress and become lethal. In people with NF1, MPNST often develops within benign tumors, especially atypical benign tumors.
The hold-up in the field
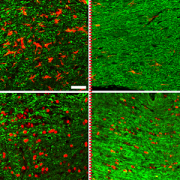
Scientists have been looking at the cell signaling process within both pre-cancerous tumors and cancerous MPNST. Previous research has shown that the MEK and MDM2 signaling pathway influence the development and growth of these tumors. By blocking this interplay, the Zhu Laboratory at Children’s National has demonstrated that tumors can get smaller when treated with drugs that inhibit MEK and MDM2 in pre-clinical models.
What’s unique
The trial is uniquely designed to evaluate target inhibition of novel drugs by looking at signals that may help in determining tumor response. This work will provide people with NF1 and MPNST potentially helpful treatments and increase the knowledge for all people with NF1 and those at risk of MPNST. The drugs will be available to all patients who enroll to the study.